 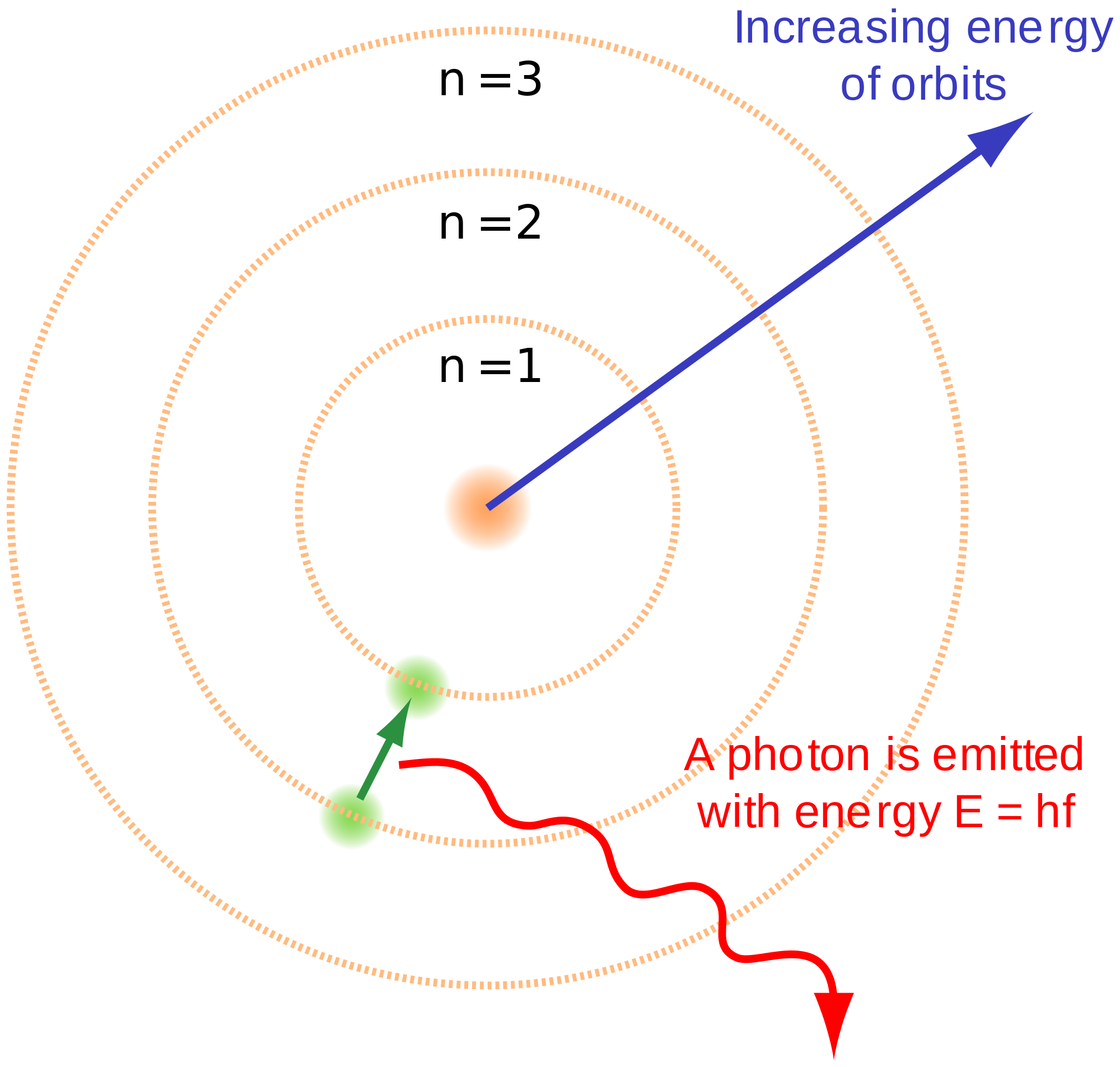
Glimpses of the "peculiar sociology" of a research institution, half of whose staff were women employed on grossly unequal terms with men, are given in the course of the narrative. The subatomic particles in the Bohr model of the atom are all. Our examination of the discovery of the Pickering series relied on the records held at the Center for Astrophysics $\vert$ Harvard \& Smithsonian (the successor institution to HCO), especially the Notebooks and Diaries of Williamina Fleming and others as well as on the Center's Glass Plate Collection. The Bohr Model of the atom was a slightly more intuitive model that human language used to describe (however, it was also inaccurate). The series of spectral lines associated with Pickering's name played a unique role on the path to quantum mechanics by serving as a proving ground for Bohr's model of the atom. Fleming was one of HCO's "computers" and the future Curator of the Astronomical Photographic Glass Plate Collection. The Bohr model (named after Danish physicist Niels Bohr) of an atom has a small, positively charged central nucleus and electrons orbiting in at specific fixed distances from the nucleus. Its spectrum has become known as the Pickering Series, even though Pickering credited Williamina Fleming (1857-1911) for the discovery. It does introduce several important features of all models used to describe the distribution of electrons in an atom. Therefore, while the Law of Definite Proportions can be used to compare two experiments in which hydrogen and oxygen react to form water, the Law of Definite Proportions can not be used to compare one experiment in which hydrogen and oxygen react to form water, and another experiment in which hydrogen and oxygen react to form hydrogen peroxide (peroxide is another material that can be made from hydrogen and oxygen).Download a PDF of the paper titled The Spectrum of He$^+$ as a Proving Ground for Bohr's Model of the Atom: A Legacy of Williamina Fleming's Astrophysical Discovery, by Maria McEachern and Bretislav Friedrich Download PDF HTML (experimental) Abstract:In 1896, Edward Charles Pickering (1846-1919), Director of the Harvard College Observatory (HCO), reported in a trio of publications the observation of "peculiar spectra" of the southern star $\zeta$ Puppis, which he attributed to an "element not yet found in other stars or on earth." Supported by laboratory spectra obtained by Alfred Fowler (1868-1940), Niels Bohr (1885-1962) showed in 1913 that this "element" was in fact ionized helium, He$^+$. Bohr’s model of the hydrogen atom provides insight into the behavior of matter at the microscopic level, but it does not account for electronelectron interactions in atoms with more than one electron. 
Bohr proposed that electrons do not radiate energy as they orbit the nucleus, but exist in states of constant energy that he called stationary states. The Law of Definite Proportions applies when elements are reacted together to form the same product. Following the discoveries of hydrogen emission spectra and the photoelectric effect, the Danish physicist Niels Bohr (1885-1962) proposed a new model of the atom in 1915. If the spheres touch, they are part of a single unit of a compound. 4 ), is a fundamental concept that states that all elements are composed of atoms. The purple spheres represent atoms of another element. The modern atomic theory, proposed about 1803 by the English chemist John Dalton (Figure 1.5.4 1.5. The electrons orbit around the nucleus similar to how planets orbit around the sun. The model is also referred to as the planetary model of an atom. In this model, the electrons travel around the nucleus of an atom in distinct circular orbits, or shells. 
Law of Definite Proportions states that in a given type of chemical substance, the elements are always combined in the same proportions by mass. In the following drawing, the green spheres represent atoms of a certain element. The model was proposed by physicist Niels Bohr in 1913. Niels Bohr - Atomic Theory, Quantum Mechanics, Nobel Prize: After the discovery of fission, Bohr was acutely aware of the theoretical possibility of making an atomic bomb. Similarly, when 2 grams of A react with 16 grams of B, they must produce 18 grams of C. The first five atomic orbitals: 1s, 2s, 2px, 2py, and 2pz. These patterns are referred to as atomic orbitals and come in a variety of shapes (basic spheres, rings, dumbbells, etc.) and the nucleus is always at the center. If 1 gram of A reacts with 8 grams of B, then by the Law of Conservation of Mass, they must produce 9 grams of C. This refuted the Bohr model of the atom, and replaced it with a pattern of probabilities as to where electrons are located around the nucleus. \): If 1 gram of A reacts with 8 grams of B, then by the Law of Definite Proportions, 2 grams of A must react with 16 grams of B.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
